Experimental performance of the IDA electrode, with a small width (3 µm and 2 µm) is compared with the 10 um width IDA electrode.
Also, the collection efficiency was compared for the three types of the IDA electrodes.
- IDA Electrode 3 µm
- IDA Electrode 2 µm
More fine IDA (Pt/Au) Electrode has been developed. Its 3 µm pitch working electrodes significantly increase redox cycle and allow
researchers to perform ultra-sensitive measurement.
IDA Au (3 µm)
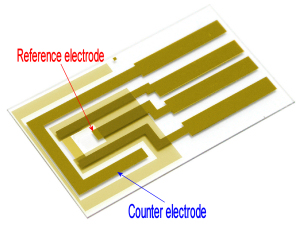
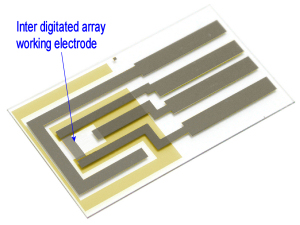
We recommend to apply Ag/AgCl ink to a reference electrode in order to stabilize potential of the electrode.
Technical data concerning IDA Electrode 3 µm
Fig. 1 -- Accuracy
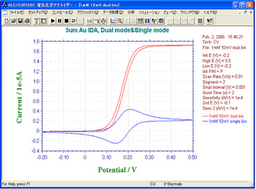
A CV curve obtained by 3 µm IDA Electrode Au [A] mirrored that of 3 µm IDA Electrode Au [B], which means both of the IDA Electrodes were produced very accurately.
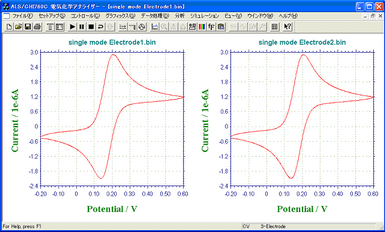

- Left: 3 µm IDA Electrode Au [A]; Right: 3 µm IDA Electrode Au [B]
- Sample: ferrocene methanol (1 mM/ 0.5 M NaCl solution)
- Mode: Single
- Sweep Rate: 30 mV/s
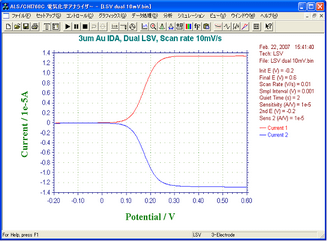
Fig. 2 -- CV by Dual mode
- Mode: Dual
- Sweep Rate at G-Electrodes: 10 mV/s
- Applied Voltage to C-Electrodes: -0.2 V
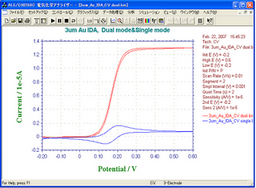
Fig. 3 & 4 -- Comparison of 3 µm IDA Au with 10 µm by Dual Mode
For 3 µm electrode, the limiting current of ferrocene methanol by CV Dual mode was 8 times as much as of the peak current by Single mode, while this ratio for 10 µm electrode was about 4. This means large increase of the redox cycle.
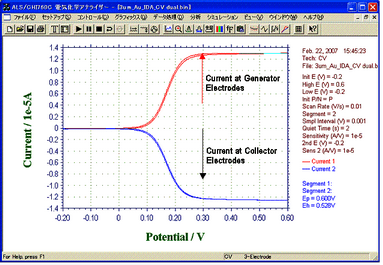
Fig. 5 -- Capture Efficiency
Figure 5 shows a result of LSV Dual mode measurement. Its collection efficiency was proved to be 96.4% by our calculation from limiting current of Collector electrodes and Generator electrodes.
- Method: LSV (Linear Sweep Voltammetry)
- Mode: Dual
- Sample: ferrocene methanol (1 mM/0.5 M NaCl solution)
- Sweep Rate: 10 mV/s
Besides CV and LSV electrochemical techniques, our IDA electrode is available for various electrochemical measuring techniques such as chronoamperometry, normal pulse voltammetry, differential pulse voltammetry etc.
Researchers can obtain more insights by using IDA electrodes.
More fine IDA (Pt/Au) Electrode has been developed. Its 2 µm pitch working electrodes significantly increase redox cycle and allow
researchers to perform high-ultra-sensitive measurement.
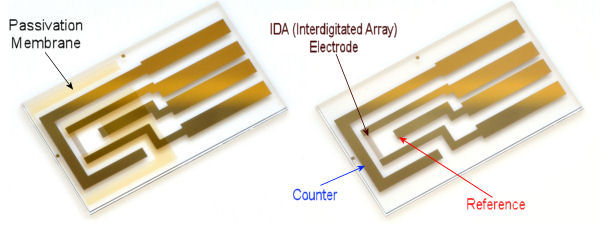
Technical data concerning IDA Electrode 2 µm
A CV curve obtained by 2 µm IDA Electrode Au.
CV by Single mode
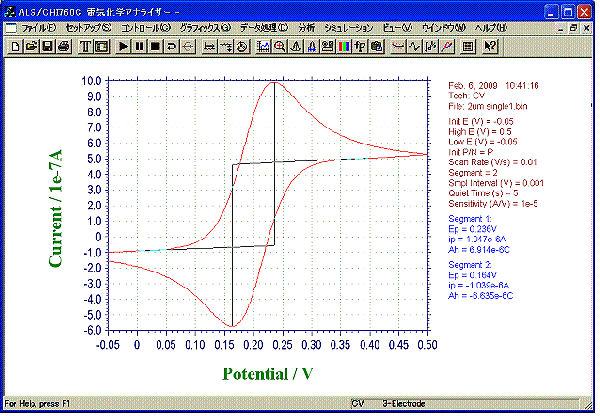
- Sample: 1 mM ferrocene methanol (0.5 M NaCl solution)
- Mode: Single
- Sweep Rate: 10 mV/s
CV by Dual mode
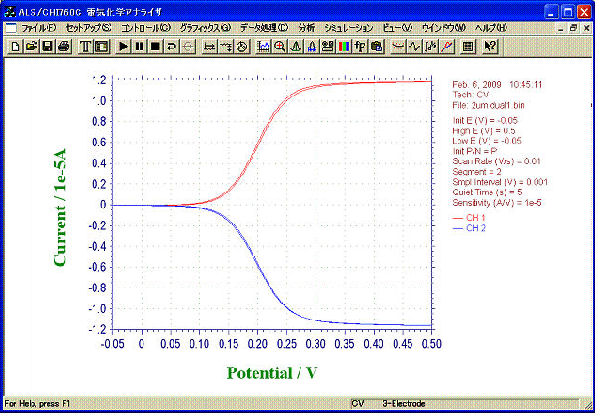
- Sample: 1 mM ferrocene methanol (0.5 M NaCl solution)
- Mode: Dual
- Sweep Rate at G-Electrodes: 10 mV/s
- Applied Voltage to C-Electrodes: -0.05 V
Its collection efficiency was proved to be 97% by our calculation from limiting current of Collector electrodes and Generator electrodes.
Capture Efficiency
Compared with IDA Au 10 µm and 3 µm, the capture efficiency of the 2 µm is higher, 93% for 10 µm and 95% for 3 µm.
For 2 µm electrode, the limiting current of ferrocene
methanol by CV Dual mode was 11 times as much as of the peak current by
Single mode, while this ratio for 10 µm and 3 µm electrode were about 4 and 7.5, respectively. This means large increase of the redox cycle.
IDA electrode line up
| Catalog No. | Description | Width (µm) | Interval (µm) | Length (mm) | Number of feet (pairs) | Film thickness |
| 012125 | IDA electrode (Au) | 10 | 5 | 2 | 65 | 90 nm∗ |
| 012126 | IDA electrode (Pt) | 10 | 5 | 2 | 65 | 90 nm∗ |
| 012128 | IDA electrode (ITO) | 10 | 5 | 2 | 65 | 100 +/- 20 nm |
| 012129 | IDA electrode (Au) | 3 | 3 | 2 | 65 | 90 nm∗ |
| 012130 | IDA electrode (Pt) | 3 | 3 | 2 | 65 | 90 nm∗ |
| 012257 | IDA electrode (Au) | 2 | 2 | 2 | 65 | 90 nm∗ |
| 012258 | IDA electrode (Pt) | 2 | 2 | 2 | 65 | 90 nm∗ |
| w/o passivation membrane | ||||||
| 012259 | IDA electrode (Au) without passivation membrane | 10 | 5 | 2.5 | 65 | 90 nm∗ |
| 012262 | IDA electrode (Pt) without passivation membrane | 10 | 5 | 2.5 | 65 | 90 nm∗ |
| 012266 | IDA electrode (Carbon) without passivation membrane | 10 | 5 | 2.5 | 65 | 1.2 +/- 0.1 µm |
| 012265 | IDA electrode (ITO) without passivation membrane | 10 | 5 | 2.5 | 65 | 100 +/- 20 nm |
| 012260 | IDA electrode (Au) without passivation membrane | 3 | 3 | 2.5 | 65 | 90 nm∗ |
| 012263 | IDA electrode (Pt) without passivation membrane | 3 | 3 | 2.5 | 65 | 90 nm∗ |
| 012261 | IDA electrode (Au) without passivation membrane | 2 | 2 | 2.5 | 65 | 90 nm∗ |
| 012264 | IDA electrode (Pt) without passivation membrane | 2 | 2 | 2.5 | 65 | 90 nm∗ |
| OPTION | ||||||
| 011066 | Cable kit for IDA electrode | |||||
| 011464 | Ag/AgCl Ink for reference electrode (2.0 mL) | |||||
∗ For Au and Pt, the thickness of the titanium adhesive layer is about 10 nm, resulting in a total thickness of 100 nm.~