Cyclic Voltammetric Stripping (CVS)
- What is CVS?
- Effects of Additives
- Voltammogram
- Influence of Chlorine ion

Cyclic Voltammetric Stripping (CVS) is one of effective electrochemical measuring techniques
to obtain ideal stripping situation by utilizing Rotating Disk Electrode (RDE).
This technique scans the potential at the working electrode which is immersed in the plating solution
together with reference and counter electrodes through an electrochemical analyzer.
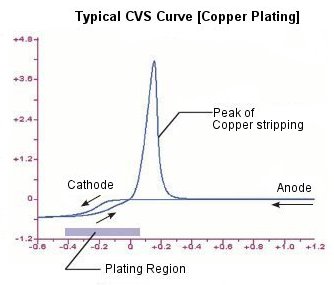
In CVS technique, working electrode can be both of anode and cathode depends on the scan direction. When the working electrode is anode, the current becomes posivite, cathode working electrode makes the current negative.
Oxidation reaction, removal of electrons from molecules or iones, occurs at anode in the electrocheical cell. On the other hand, reduction reaction, addition of electrons to molecules or iones, occurs at cathode. The potential is plotted on the X-axis and the current is shown on the Y-axis.
In CVS for copper plating, deposition occurs in reduction region. Reversal of the potential from reaction potential to oxidation potential triggers desorption of the copper.
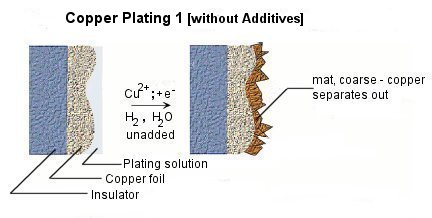
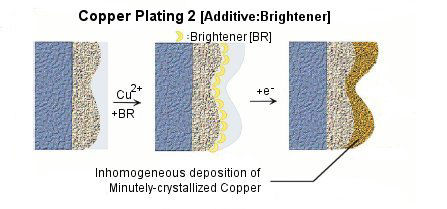
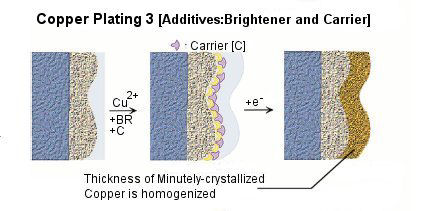
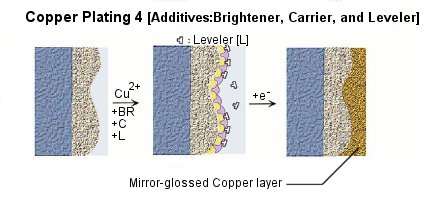
Various kinds of additives are applied into plating solution for the purposes of miniaturization of crystal grain, lustering, leveling, and homogenization of the film thickness etc. Choice, combination, and density control of additives play important roles to improve the performance and the quality of plating.
Because the precipitate structure of copper is very unstable, additives such as brightener, carrier, and leveler are added into the plating bath for the above purposes. Figure shows the effects on copper precipitation of each additives.
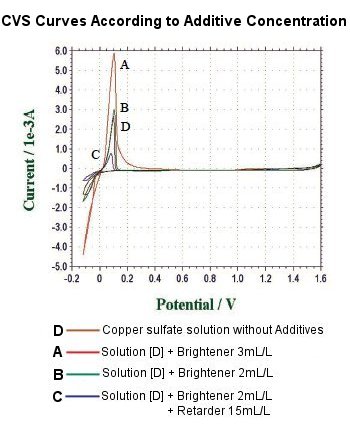
Real-time measured voltammogram contains essential information to learn the condition of plating bath. Figure shows the voltammograms measured :
A - copper sulfate solution + brightener 3 mL/L
B - copper sulfate solution + brightener 2 mL/L
C - copper sulfate solution + brightener 2 mL/L + retarder 15 mL/L
D - copper sulfate solution without additives
Quantity of Coulomb is obviously curbed by adding brightener and retarder. Measuring brightener and retarder in plating solution with acculacy becomes significant to control the condition of metal plating.
In CVS, quantity of Coulomb is proportional to the quantity of copper which separates out at the working electrode. Especially the quantity of the coulomb at the peak of copper removing means effective activity of the additive, rather than concentration level.
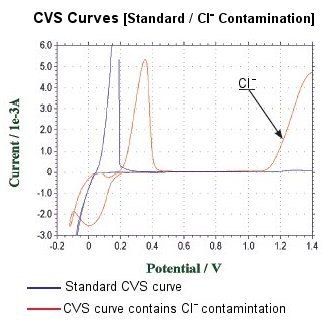
CVS measurement is very much effective to obtain not only general function, but also accurate information in the plating bath.
When the chlorine ionic contamination occurs, the voltammogram shows a distinguishing waveform at anode side.
When the quantity of the plate is measured, the area of copper stripping peak equals the quantity of electrical charge. This quantity is automatically calculated on the software. Calculate the proportion of the peak area of the solution with additions (Ar) to the area of reference solution (As). Thus CVS enables to obtain the optimal plating condition.
Improve the Quality of through-hole plating
To achieve homogeneous plate thickness has great importance in order to reduce defect in the through-hole of circuit board. CVS helps in decision of optimum parameter for high-density plating.