Introduction to Current and Reaction Rate
This technical note section will introduce the equilibrium electrode potential, the meaning of polarization and overpotential in electrode reaction kinetics, the relationship between current and potential, the meaning of the standard rate constant, the influence of mass transfer processes, the relationship between current and substance concentration, the reversibility of electrode reactions, and electrode processes involving homogeneous chemical reactions. Finally, it will introduce the derivation of the Butler-Volmer equation in electrode reaction theory, the high overpotential approximation and the low overpotential approximation, and the relationship between mass transfer and electrode reactions in electrolytes.
- Part I: Electrode Potential Equilibrium and Electrode Reaction Kinetics (1)
- Part II: Electrode Potential Equilibrium and Electrode Reaction Kinetics (2)
Part I: Electrode Potential Equilibrium and Electrode Reaction Kinetics (1)
♦ Balance electrode potential
When an electrochemical reaction system is in equilibrium, even if no apparent current flow is observed, this does not mean the reaction has completely stopped. From a kinetic perspective, this represents a state of dynamic equilibrium where the forward reaction rate equals the reverse reaction rate. Under these conditions, the terminal voltage (i.e., electromotive force) stabilizes at a constant value. This implies that the potentials of the two electrode pairs on either side remain fixed at a specific level. This potential is termed the equilibrium electrode potential, or simply the equilibrium potential.
The equilibrium potential Eeq of a simple electron transfer reaction occurring at a single electrode (Equation 1) can be quantitatively expressed using the Nernst equation (Equation 2) based on the concentrations c (strictly speaking, the activities a) of the oxidized and reduced species present in the electrolyte solution.

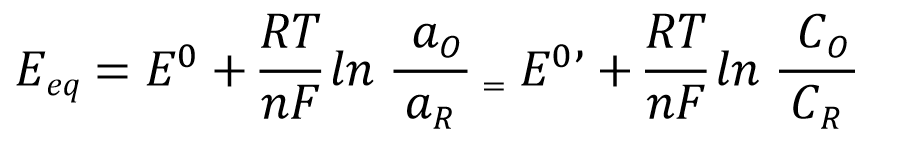
F:
R:
T:
E0:
E0':
Eeq:
Faraday's constant (96485 Cmol-1)
Ideal gas constant (8.31J K-1 mol-1)
Absolute temperature (K)
Standard electrode potential (standard electrode potential),
aR = aO = 1 (Standard temperature) Equilibrium potential at the time
Formal potential or conditional potential,
the standard electrode potential based on concentration.
Equilibrium potential Eeq (The experimental quantity that varies
according to the concentration ratio of redox substances participating in the reaction).
Therefore, the equilibrium potential Eeq is an experimental quantity that can vary depending on the concentration ratio of the redox species involved in the reaction. As previously explained, the electromotive force of a galvanic cell can be calculated using the Nernst equation from the quantitative relationships of these electroactive species.
♦ Electrode Reaction Kinetics (1): Polarization and Overpotential
When equilibrium is disrupted and the reaction begins to proceed in one direction, this direction can be reflected in the sign of the current (oxidation currents are positive, reduction currents are negative). For example, for reactions where oxidation is the positive reaction, the net reaction rate V can be expressed as the oxidation reaction rate minus the reduction reaction rate (Eq. 3).

Here, each reaction rate can be expressed as the product of the reactant concentration (mol cm-3) and the reaction rate constant k (cm s-1), just as in homogeneous reaction systems. Thus, the forward reaction rate Vox and the reverse reaction rate Vred can be represented by Eq. 4 and Eq. 5 below.

It should be noted that the CRs and COs appearing here do not refer to the concentration within the solution itself, but rather to the concentration at the interface between the electrode and the solution. This is hereafter referred to as surface concentration.
Because the absolute value of the current is directly proportional to the reaction rate. The oxidation-reduction current and reaction rate can be quantitatively related through Equations 6 and 7, and the observed net current I can be expressed as shown in Equation 8.
When a reaction occurs and current flows, the electrode potential begins to shift from its previous equilibrium value according to the direction and rate of the reaction. In other words, “the potential shift is a result of the reaction occurring,” but conversely, “forcing the potential away from its equilibrium value can enable the reaction to proceed.”
Potential or current, one of these can be used as a control variable and actively altered to track how the other quantity responds to this change, thereby providing a theoretical explanation. This constitutes the fundamental approach of electrochemical measurement methods.
Incidentally, both the operation of “shifting the potential from the equilibrium potential to induce a reaction” and the phenomenon of “potential shift caused by the reaction” are described using the electrochemical term “polarization.” (The amount of deviation from the equilibrium potential is referred to as overvoltage or overpotential.)
Its meaning is that “to generate a current of a certain magnitude, a voltage higher than the equilibrium potential is required.”
When the equilibrium potential is denoted by Eeq, the overvoltage (symbolized by the Greek letter eta) is the quantity defined by Eq. 9.
Where E is the electrode potential. Figure 1 shows the relationship between overvoltage and current.

Fig. 1 Relationship between Overpotential (Overpotential) and Current.
In summary, whenever a non-zero current is observed, regardless of its magnitude, an overpotential (or overpotential) is generated.
Part II: Electrode Potential Equilibrium and Electrode Reaction Kinetics (2)
♦ Relationship between current and potential
We know that an overpotential is required to drive a reaction in a given direction. So, what is the relationship between the magnitude of the overpotential and the actually observed current?
As previously mentioned, current is an indicator of reaction rate. On the other hand, the electrode potential can be said to play a role in regulating the energy balance of the reaction. In homogeneous chemical reactions, whether a reaction is exothermic or endothermic is determined by the energy relationship between the reactant system and the product system. Similarly, in electrochemical reactions, whether energy can be delivered to the outside (as in a battery) or must be supplied from the outside (as in electrolysis) is determined by the energy difference between the reactant system and the product system. Here, let us consider the influence of electrode potential on electrode reactions from an energy perspective.
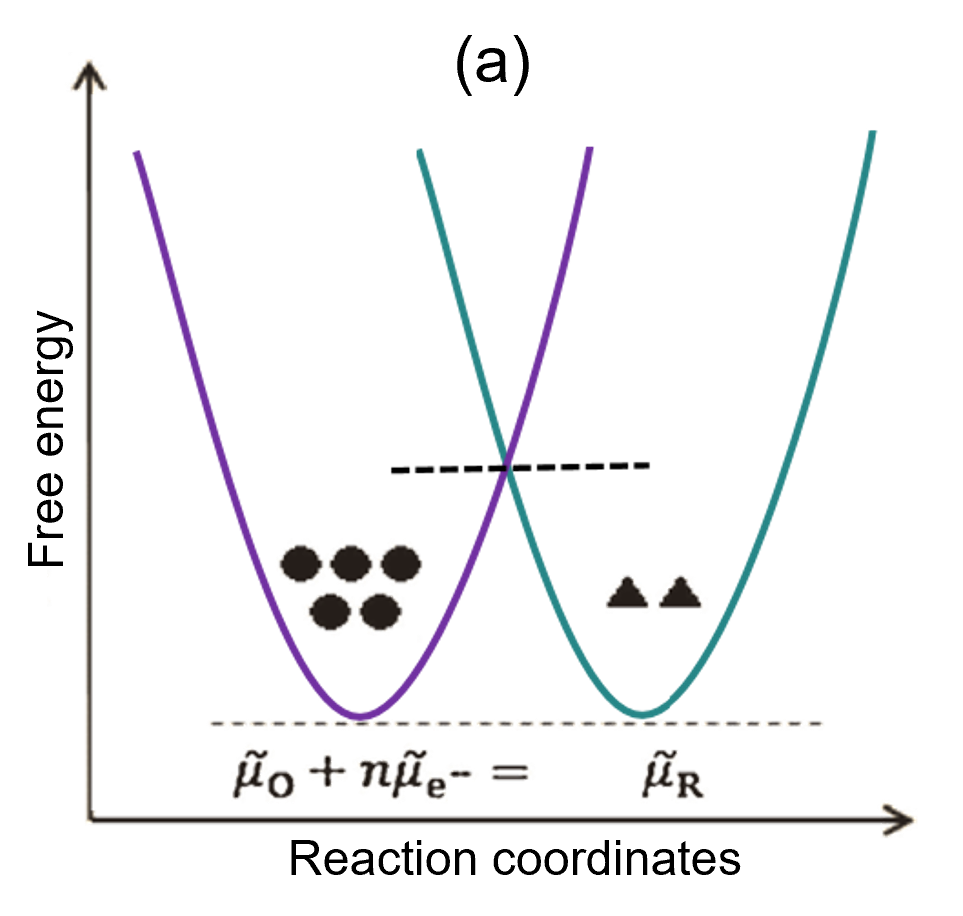
(a) Initial state (equilibrium state): In the equilibrium state, the free energies of the reactant system and the product system are equal. Obviously, the reaction does not proceed in either direction, so I = 0. The symbols ● and ▲ represent the relative concentrations of O and R, respectively. Note that equilibrium does not imply equal concentrations.
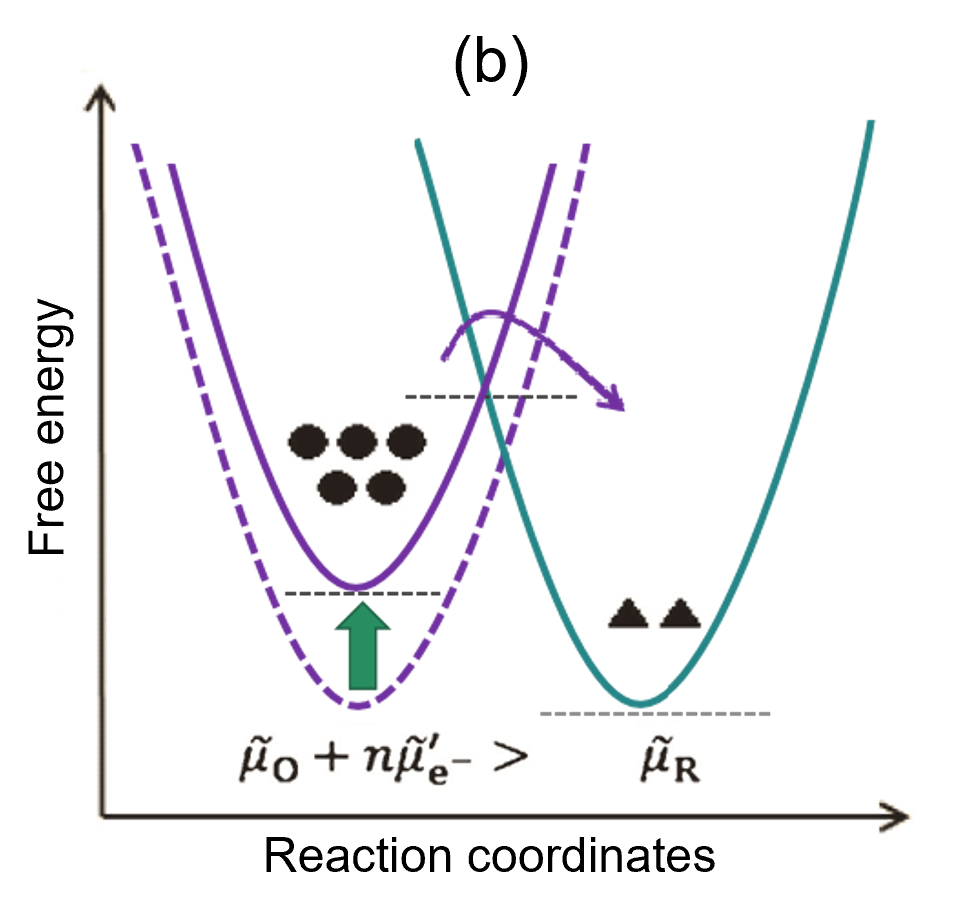
(b) When a negative overpotential is applied to the state of (a), the energy of the electrons in the electrode increases, the free energy of the reaction system increases temporarily, the activation energy of the reaction in the O → R direction and the R → O direction changes, and the reaction proceeds to the right side to produce a current flow (I < 0)
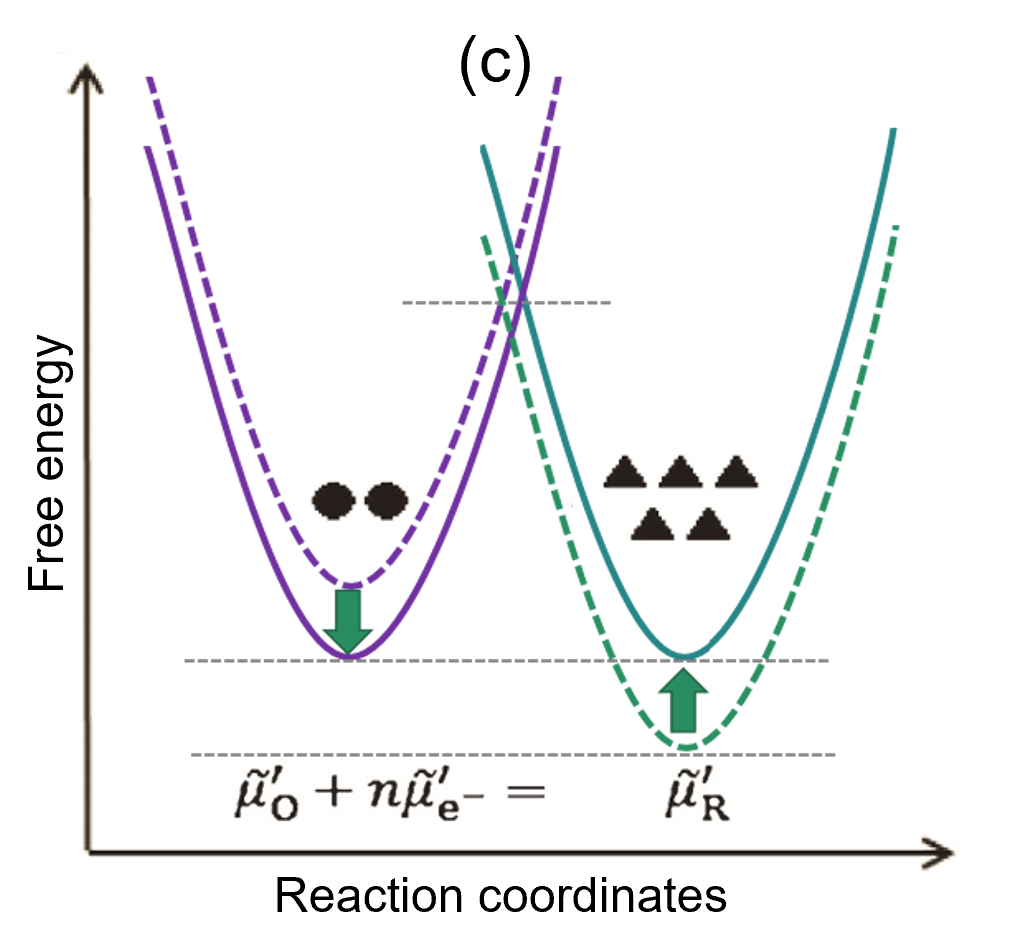
(c) Final state (equilibrium state): As the reaction progresses, the free energy difference between the reaction system and the generated system becomes smaller and smaller. At the same time, the difference in activation energy is eliminated, so the current decreases, and the system reaches a new equilibrium when I = 0.
If the initial state is at equilibrium, the free energies of the two systems are equal (Fig. 2(a)). In this state, when a negative overpotential is applied to the electrode, the energy of electrons inside the electrode increases, and the free energy of the reaction system increases. The free energy of the reaction system shows a temporary rise; as a result, the activation energy for the reduction reaction in the O → R direction becomes smaller than before, so the reaction tends to proceed to the right, generating a flow of current (I < 0). At the same time, the activation energy for the opposite oxidation reaction in the R → O direction increases (Fig. 2(b)). As O is consumed and R increases, the energy of each system also changes, and after a long period of time they eventually become equal. At this point, the reaction has clearly stopped, and the system reaches a new equilibrium state (Fig. 2(c)).
From the above, it can be seen that a current can be observed when the system is in an energetically unstable nonequilibrium state (transition state).
A unique feature of electrochemical reactions is that the direction of the reaction can be controlled by the potential applied to the electrode.
When a positive overpotential opposite in sign to the previous one is applied to the electrode, the opposite phenomenon occurs: the reaction proceeds from right to left, that is, an oxidation reaction. In other words, the relative magnitudes of the activation energies are reversed depending on the sign of the applied overpotential.
Similar to conventional chemical reactions, the rate constant k of electrochemical reactions varies with activation energy Ea according to the Arrhenius equation.
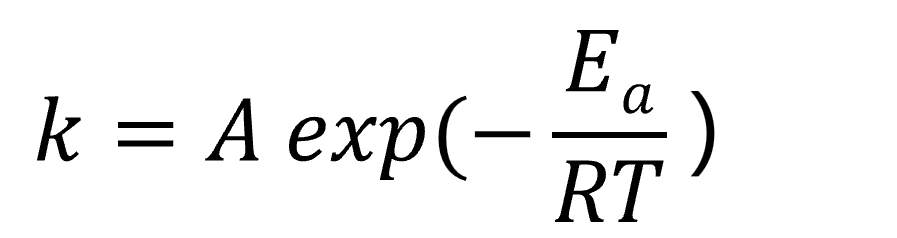
(R: gas constant, T: absolute temperature, A: coefficient)
When an overpotential η is generated, the activation energy (Ea) of the oxidation reaction and reduction reaction is expressed as follows.
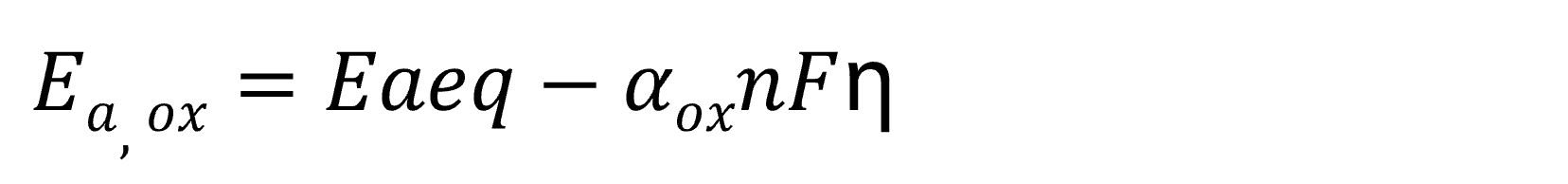
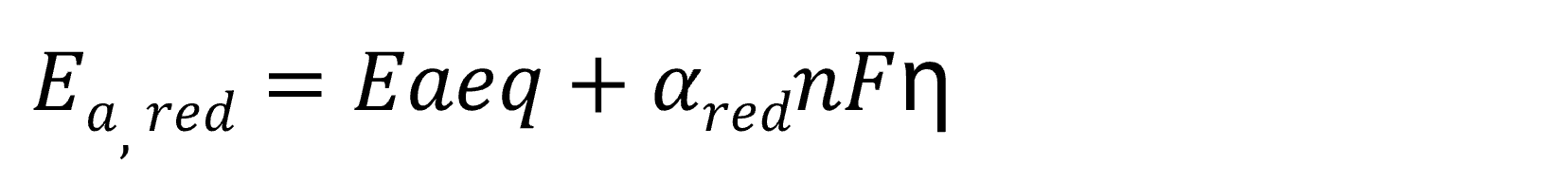
Here, Ea‚eq represents the activation energy at equilibrium potential, while α denotes the transfer coefficient. The transfer coefficients for oxidation and reduction reactions satisfy the relationship 0 < αox, αred < 1 and αox + αred = 1. (The transfer coefficient quantifies the contribution of the applied overpotential to the change in activation energy for oxidation or reduction reactions.)
Based on the above relationship, the reaction rate constants are expressed as follows.
Oxidation reaction rate constant:
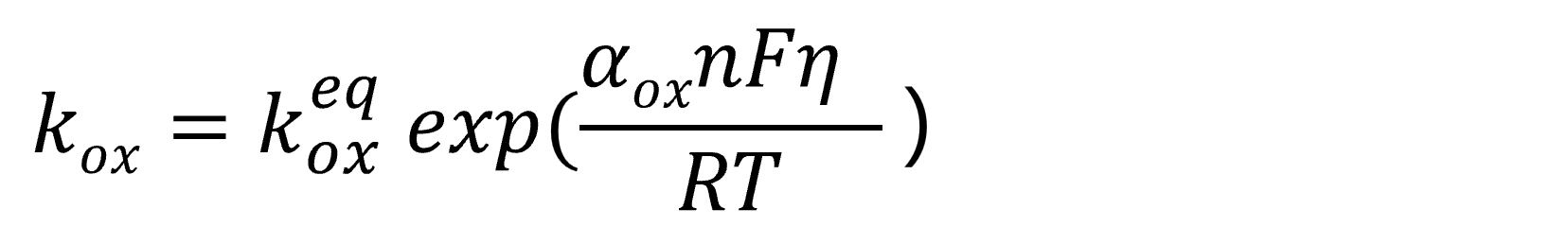
Reduction reaction rate constant:
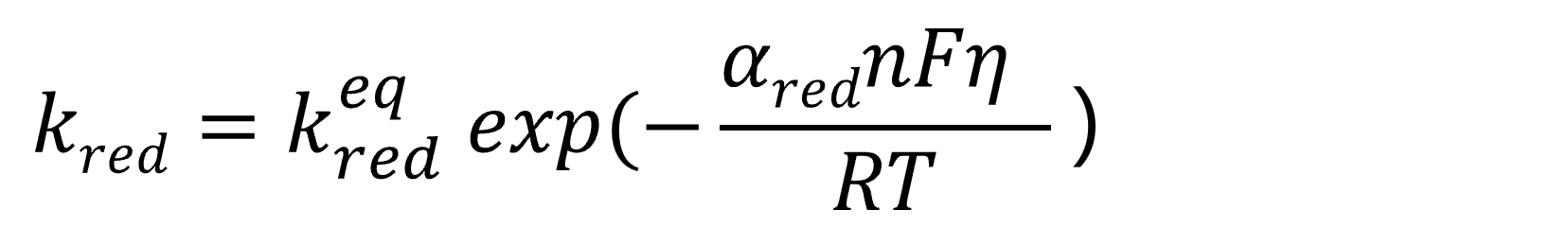
Eq. 13 represents the oxidation reaction rate constant, while Eq. 14 represents the reduction reaction rate constant. The rate constant keq denotes the reaction rate constant at the equilibrium potential. From the above equations, it can be seen that the reaction rate, i.e., the current, exhibits an exponential dependence on potential changes. This overpotential is termed the activation overvoltage (or activation overpotential) or superpotential.
The exponential relationship between the electrolysis current and the applied voltage was first discovered experimentally, and the relationship between the two was formulated in the well-known Tafel equation.
As described above, the equation that theoretically considers the relationship between activation energy and overpotential and quantitatively relates it to the reaction rate (current) is the Butler–Volmer equation.

