How to choose a reference electrode for practical measurements
M.Sc. Kimihiro Kinugasa
Contents
Chapter 1: Understanding the "Reference electrode" role in Electrochemical Measurements
Chapter 2: Recommended reference electrodes by measurement environment
Chapter 3: Practical guide: How to choose the right reference electrode
Chapter 4: Common mistakes and how to prevent them
Chapter 5: Quality and traceability supporting reliability
Chapter 6: Maintenance and troubleshooting for long-term use
Chapter 7: Summary on reference electrodes
- Chapter 1: Understanding the "Reference electrode" role in Electrochemical Measurements
- Chapter 2: Recommended reference electrodes by measurement environment
- Chapter 3: Practical guide: How to choose the right reference electrode
- Chapter 4: Common mistakes and how to prevent them
- Chapter 5: Quality and traceability supporting reliability
- Chapter 6: Maintenance and troubleshooting for long-term use
- Chapter 7: Summary on reference electrodes
Chapter 1: Understanding the "Reference electrode" role in Electrochemical Measurements
1-1 Why Reference Electrodes is used in Electrochemical Measurements?
In general, electrochemical reactions can be carried out using two electrodes: a positive electrode and a negative electrode. In practice, many electrolysis reactions are performed using such two-electrode systems.
However, when a voltage is applied between these two electrodes (the working electrode and the counter electrode), the applied potential is divided between them. As a result, while the potential difference between the two electrodes is known, the exact potential at each individual electrode cannot be determined directly. Furthermore, when electron transfer occurs in the solution due to electrolysis (i.e., when a current I flows), a potential drop (E = IR) occurs due to the resistance R of the solution, making accurate potential measurement difficult.
So how can we accurately know and control the potential of the working electrode where the reaction of interest occurs? In electrochemical measurements and organic electrosynthesis, precise control of the working electrode potential is critically important.
1-2 Standard Reference Electrode (SHE): Mechanism and Challenges
An electrode that serves as a potential reference is called a reference electrode. Before discussing reference electrodes in detail, let us first review the main functions of an electrochemical instrument known as a potentiostat. (Fig. 1-1)
- It controls the potential of the working electrode relative to the reference electrode.
- It measures the current flowing through the working electrode.
- It prevents current from flowing through the reference electrode.
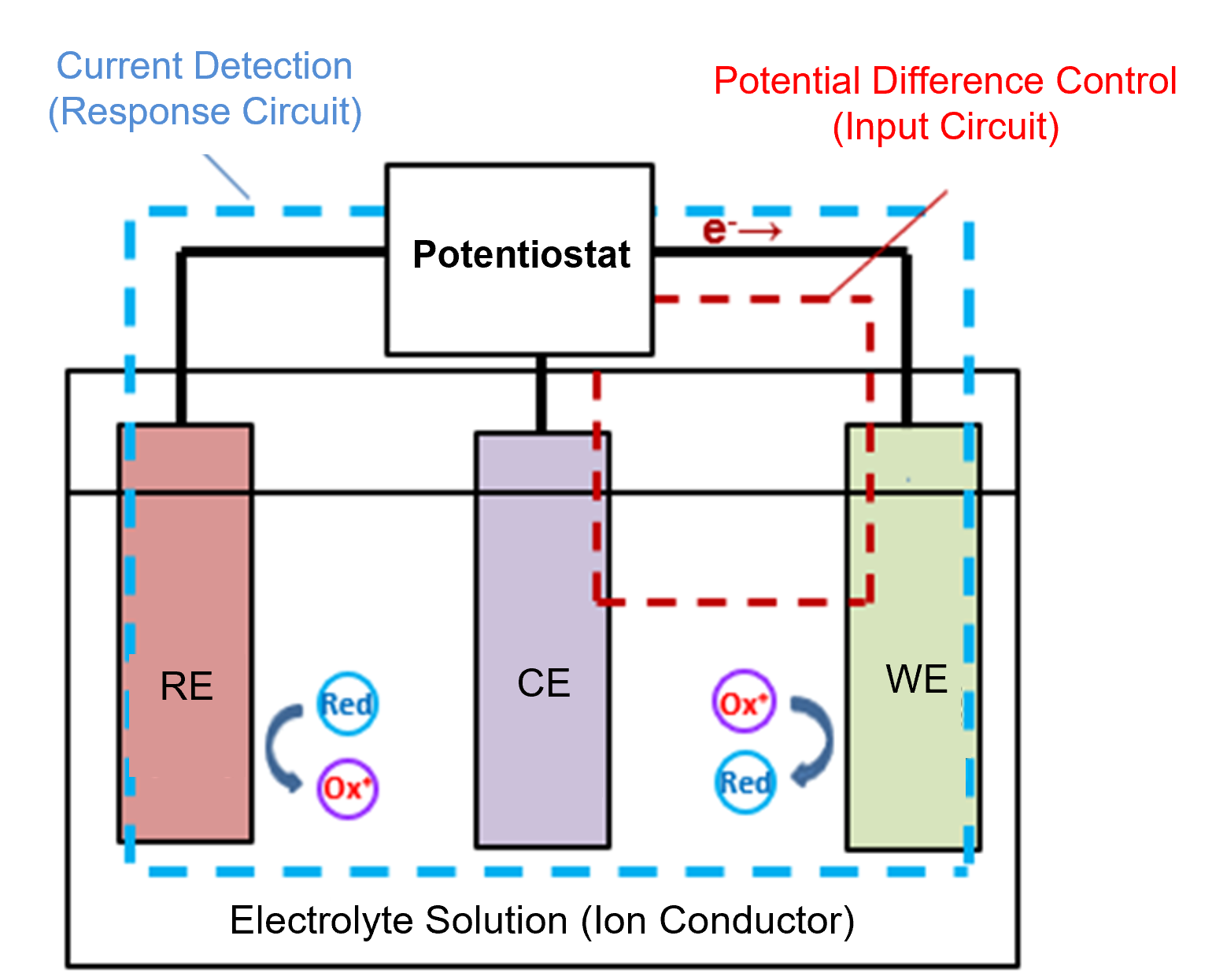
Fig. 1-1 Schematic diagram of a three-electrode configuration.
A potentiostat controls the system so that a specified potential (for example, +500 mV) is always applied to the working electrode relative to the reference electrode. Therefore, accurate control of the working electrode potential requires a reference electrode that provides a stable and universal potential.
The "Standard Hydrogen Electrode (SHE)" is proposed as the reference for this purpose. The SHE maintains a universal potential by immersing a platinum wire in a solution of pH = 0 ([aH+] = 1) and constantly bubbling hydrogen gas at 1 atm (Fig. 1-2). By using the SHE as a reference, the electrode reaction potentials of various substances can be compared against a common standard.
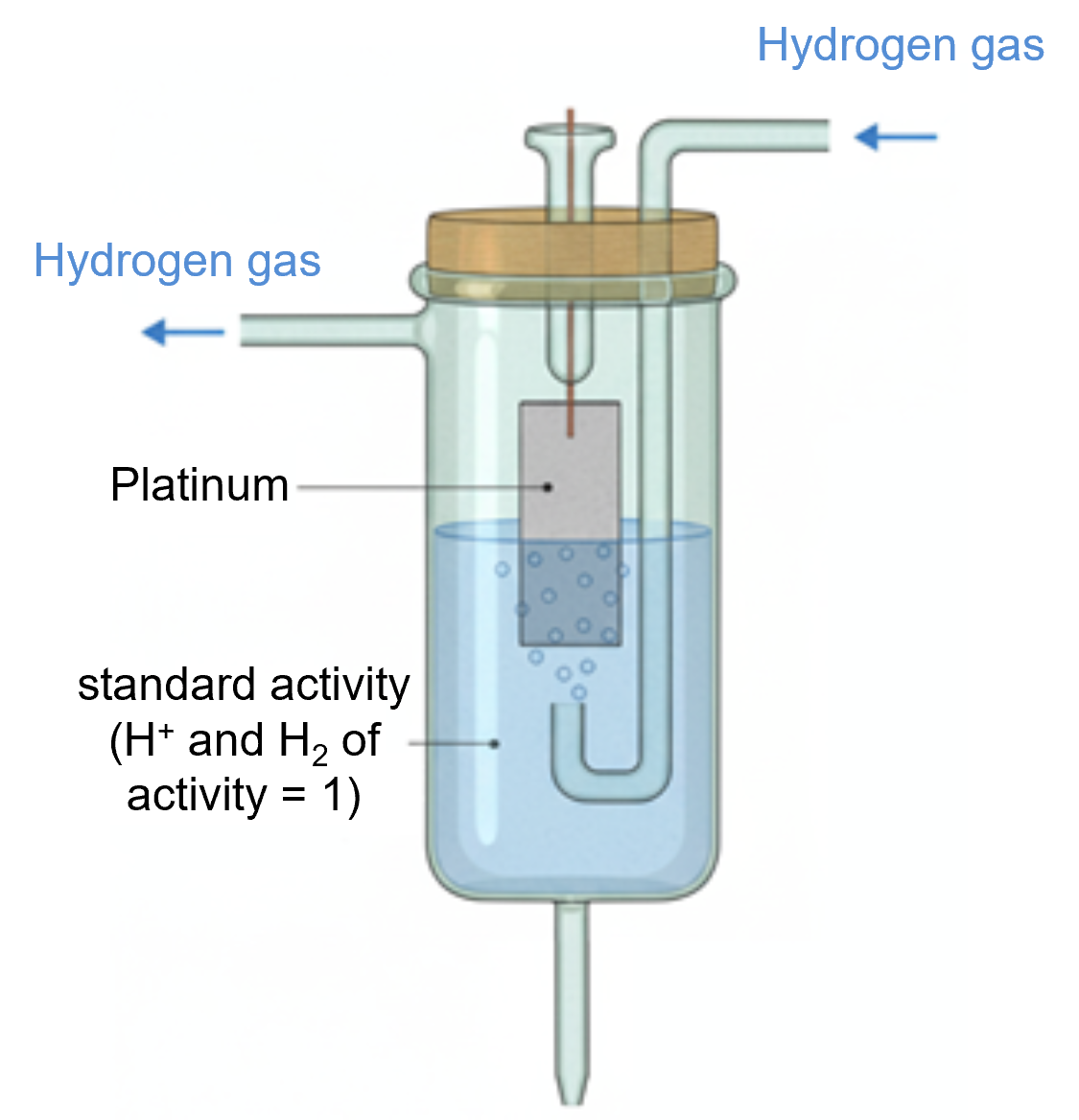
Fig. 1-2 Schematic illustration of the operation of the Standard Hydrogen Electrode (SHE).
However, SHE is difficult to operate in practice due to the need for continuous hydrogen gas supply and strict safety management, making it unsuitable for routine use.
1-3 Practical Selection of Reference Electrodes: What is a "Secondary Standard" that combines Stability and Usability?
While SHE is theoretically the most stable reference, practical measurements typically use secondary standard reference electrodes (non-polarizable electrodes) with known potentials relative to SHE.
Table 1. Comparison of typical reference electrodes:
| Electrode | Composition/Principle | Standard Potential (vs. SHE) | Main features | Typical applications |
| SHE (Standard Hydrogen Electrode) | Pt + H2 gas + H+ (a = 1) | 0.000 V | Fundamental reference, difficult to operate | Fundamental research, calibration |
| RHE (Reversible Hydrogen Electrode) | Pt + H2 + solution with same pH as sample | pH-dependent (−59 mV/pH) | No liquid junction potential; useful for pH-dependent systems | Analysis of electrode reaction mechanisms |
| Ag/AgCl electrode | Ag | AgCl | KCl (saturated) | +0.199 V (sat. KCl) |
| Calomel electrode | Hg | Hg2Cl2 | KCl (saturated) | +0.244 V |
| NaCl-type Ag/AgCl electrode | Ag | AgCl | NaCl solution | +0.196 V (3 M NaCl) |
In aqueous systems, electrodes that maintain a stable potential through equilibrium between internal oxidized and reduced species, without the use of hydrogen gas, are commonly used. Typical examples include the silver/silver chloride electrode (Ag/AgCl) and the calomel electrode. In Ag/AgCl electrodes, a reversible redox reaction, such as the one shown in Eq. 1, occurs at the silver surface.
The rightward direction represents oxidation, and the leftward direction represents reduction. Because these reactions occur equally in both directions at equilibrium, the electrode potential remains stable and suitable for use as a reference. Calomel electrodes also exhibit stable potentials, however, because they contain mercury, their use has declined due to environmental concerns. As a result, Ag/AgCl electrodes are now widely used.
BAS provides reference electrodes that use polymethylpentene resin holders for resistance to external impact, as well as ceramic liquid junctions (porous ceramics) with improved alkaline resistance. In addition to potassium chloride (KCl), BAS also offers Ag/AgCl electrodes that use sodium chloride (NaCl) as the internal solution.
This lineup allows users to select reference electrodes that maintain long-term stability depending on the measurement environment. For example, when perchloric acid is used in applications such as fuel cell catalyst evaluation, electrodes that use KCl as the internal solution may suffer clogging of the liquid junction due to precipitation of low-solubility potassium perchlorate (KClO4). In such cases, selecting a NaCl-based Ag/AgCl reference electrode (RE-1BP, aqueous reference electrode) enables stable measurements over extended periods.
There is also a reference electrode similar to SHE called the Reversible Hydrogen Electrode (RHE: Reversible Hydrogen Electrode). Like SHE, RHE uses hydrogen gas, but differs in that the pH of its internal solution is the same as that of the measurement solution. The potential of RHE follows the Nernst equation: at pH = 0 it has the same potential as SHE, but for each increase of one pH unit, the potential shifts by −59 mV relative to SHE. Although this change in potential may seem problematic, it poses no issue because the potential can be calculated accurately from the pH using the Nernst equation.
A major advantage of RHE is that, because it uses an internal solution with the same pH as the measurement solution, no liquid junction potential is generated. For reactions involving H+, the reaction potential itself depends on pH. For example, when scanning a platinum electrode in the negative direction using an Ag/AgCl reference electrode, water decomposition (hydrogen gas evolution) occurs at relatively positive potentials in acidic solutions, whereas in alkaline solutions it begins at more negative potentials. With RHE, the reference electrode potential shifts according to pH following the Nernst equation, so the potential at which water begins to decompose relative to RHE remains constant regardless of pH. Visualizing this difference can help in understanding the concept.
When measurements are performed using the RHE scale, the potentials for hydrogen evolution and oxygen reduction reactions (ORR) can be readily identified, making it easier to set the potential scan range for cyclic voltammetry (CV).
In the following chapters, in addition to the characteristics of each reference electrode introduced here, we will explain methods for selecting, checking, and maintaining the reference electrodes handled by BAS.
Chapter 2: Recommended reference electrodes by measurement environment
2-1 Aqueous solutions: Ag/AgCl, Calomel, and Alkaline reference electrode
The silver/silver chloride electrode is one of the most commonly used electrodes. BAS offers the following electrodes.
2-1-1 Silver/Silver chloride reference electrodes
RE-1B Reference electrode (Ag/AgCl) (Cat. No. 012167)
Compact reference electrode that is easy to handle, with an internal solution of 3 M NaCl (sodium chloride).
The main reason for using NaCl as the internal solution is to prevent clogging of the liquid junction under certain measurement conditions (see references 1–3), because the liquid junction is made of ion permeability porous glass, this electrode should be used primarily in acidic to neutral pH ranges.
When used in alkaline conditions, the ion permeability porous glass at the liquid junction gradually dissolves, rendering the electrode unusable.
RE-1BP Reference electrode (Ag/AgCl) (Cat. No. 013613)
A slight physical and chemical resitance silver/silver chloride reference electrode.
It uses polymethylpentene, which is more cost-effective and offers excellent chemical resistance and impact resistance, and the liquid junction is made of ceramic material, allowing the electrode to be used in environments up to mildly alkaline conditions.
RE-1CP Reference electrode (Ag/AgCl/Saturated KCl) (Cat. No. 013691)
Electrode is filled with a saturated potassium chloride (KCl) internal solution. Because the constituent ions (K+ and Cl-) have nearly identical ionic mobilities in aqueous solutions, the liquid junction potential between the electrode and the sample is minimized, making it an ideal reference electrode.
However, if the sample contains certain anions such as perchlorate (ClO4-), low-solubility potassium perchlorate (KClO4) may precipitate at the liquid junction. This can cause clogging and lead to unstable measurements, such as increased noise.
Therefore, careful consideration must be given to the composition of the sample solution when using this electrode.
2-1-2 Mercury based reference electrodes
Consists of mercury, mercury(I) chloride, and mercury(II) sulfate, offering excellent potential stability. It is an aqueous reference electrode that was widely used for routine measurements until recently as an alternative to the Standard Hydrogen Electrode (SHE), which requires hydrogen gas. However, because its composition involves mercury, its use has been declining due to environmental concerns.
RE-2BP Calomel Reference electrode (Cat. No. 013693)
Calomel type aqueous reference electrode consisting of mercury and mercury(I) chloride in contact with a platinum wire. Filled with a saturated potassium chloride (KCl) internal solution, providing a highly stable potential.
The electrode body is made of polymethylpentene resin, providing high resistance to physical impact and external shocks.
RE-2CP Reference electrode (Cat. No. 013692)
Mercury based reference electrode (a variation of the calomel electrode) uses saturated potassium sulfate (K2SO4) as the internal solution. It is suitable for analyses in which chloride ions (Cl- from KCl) must be avoided. The electrode body is made of polymethylpentene resin, providing high resistance to physical impact and external shocks.
RE-61AP Reference electrode for alkaline solution (Cat. No. 013694)
This electrode is a type of calomel reference electrode developed primarily for use in alkaline solutions. The electrode reaction is as follows:
Remove the screw-type cap at the top and fill the electrode with an alkaline solution before use (the internal solution is not included with the product).
After filling, immerse the electrode in the same solution as the internal solution for approximately one day prior to use.
2-2 Non-aqueous solutions: Silver/Silver ion electrode and IUPAC recommended corrections
2-2-1 Silver/Silver ion (Ag/Ag+) reference electrodes
The most commonly used reference electrode for nonaqueous solvent systems is the Ag/Ag+ reference electrode.
It is prepared by filling a glass tube with the same solvent as the sample solution, containing Ag+ ions and a supporting electrolyte at a concentration of approximately 0.1 M, and immersing a silver wire in the solution.
RE-7N Non Aqueous reference electrode (Cat. No. 013848)
BAS non-aqueous reference electrodes for electrochemical measurements are silver/silver ion (Ag/Ag+) types and are supplied as an assembly kit (solvent is not included in the kit).
A typical example of an internal solution is 0.01 M silver nitrate (AgNO3) and 0.1 M tetrabutylammonium perchlorate (TBAP) in acetonitrile. When the internal solution is prepared to match specific measurement requirements, this electrode can be used for a wide variety of experimental setups.
By using the 6 mm diameter sample holder (Cat. No. 012176), it is also possible to fabricate custom reference electrodes for different solvents. While this allows for flexible combinations of solvents and supporting electrolytes, it is important to note that in non-aqueous systems, the reference electrode potential can vary significantly depending on the solvent used.
2-2-2 Setting the potential references (IUPAC recommended methods)
In non-aqueous systems, the reference electrode potential can vary greatly depending on the solvent. Therefore, the use of an internal standard substance as an absolute potential reference is recommended.
IUPAC Recommended standard:
The International Union of Pure and Applied Chemistry (IUPAC) recommends adding ferrocene (or bis(biphenyl)chromium salts) to the measurement solution at the end of the experiment, and determining its redox potential from the resulting cyclic voltammogram, which is then used as the reference (zero point). Because the redox potential of ferrocene is minimally affected by the solvent effects, it is widely used as a potential reference in non-aqueous electrochemical measurements.
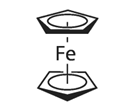
Fig. 2-1 Structure of ferrocene
2-3 Reactions involving Protons (H+): Advantages of the Reversible Hydrogen Electrode (RHE)
RHEK Reversible hydrogen electrode kit (Cat. No. 013597)
The reversible hydrogen electrode (RHE) was discussed in the Section 1–3. It is an extremely important reference electrode for studying electrochemical reactions involving protons (H+), as it allows for the elimination of the pH dependence of the electrode potential.
BAS offers a "Reversible Hydrogen Electrode Kit" (RHEK), which utilizes hydrogen gas generated by the electrolysis of a strong acid. The kit is designed to store hydrogen gas, eliminating the need for a continuous gas flow and making it exceptionally easy to handle.
In addition, BAS offers a H2G1 Portable Hydrogen Generator, which allows for the easy filling of the RHEK with hydrogen gas even without the use of a potentiostat.
Chapter 3: Practical guide: How to choose the right reference electrode
3-1 Selection criteria for aqueous and non aqueous reference electrode: Liquid junction potentials and supporting electrolytes
In Chapter 2, the characteristics of various reference electrodes were explained. When selecting a reference electrode for actual measurements, please refer to the following points.
- A reference electrode must exhibit a stable and constant potential over long periods of time. The stability of the electrode potential is maintained by the internal redox species being in a state of equilibrium.
- Minimize liquid junction potentials between the measurement solution and the internal solution of the reference electrode. To achieve this, high concentrations of salts with similar ionic mobilities, such as potassium chloride (KCl) or potassium nitrate (KNO3), are typically used as the internal solution.
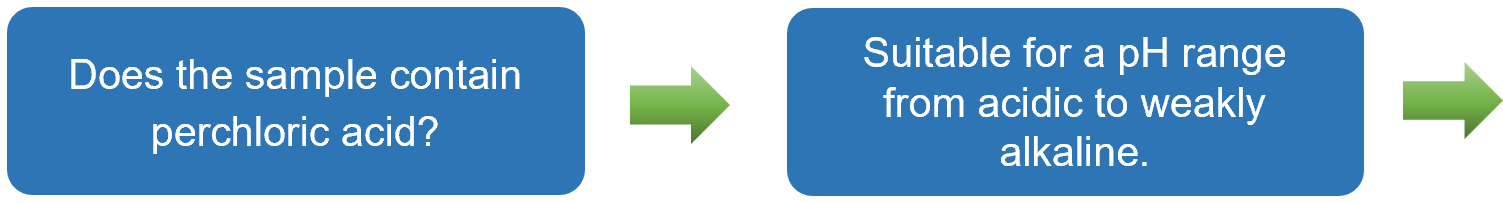
- If perchlorate ions (ClO4-) are present as anions in the sample solution, select the NaCl-based RE-1BP reference electrode.
- When investigating electrode reactions involving H⁺ ions (e.g., fuel cell catalyst evaluation), selecting a Reversible Hydrogen Electrode (RHE) is highly advantageous. Because the reference potential shifts in accordance with the solution pH, the pH dependence of the electrode potential and that of the RHE cancel each other out. This allows overpotentials to be obtained directly, making data analysis extremely straightforward.
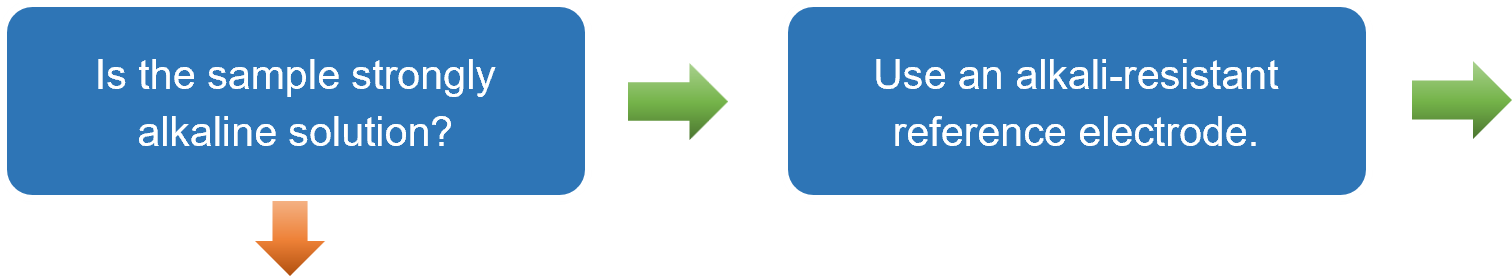
- For measurements in strongly alkaline solutions, select the RE-61AP alkaline reference electrode. If the liquid junction uses ion-permeable glass, the glass may dissolve under strong alkaline conditions. In such cases, countermeasures such as using electrodes with ceramic junctions are necessary.
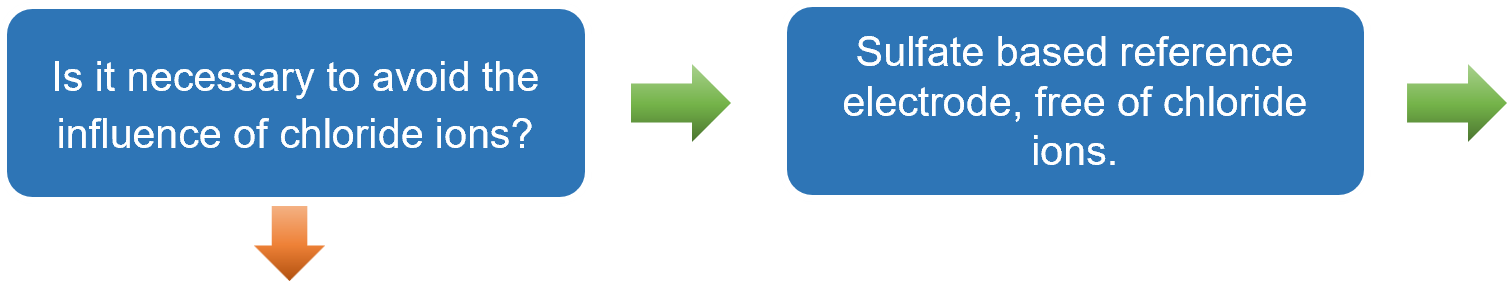
- If contamination by chloride ions must be avoided, use the RE-2CP reference electrode.
3-2 Selection and preparation for non aqueous (organic solvent)
- Use the silver/silver ion (Ag/Ag⁺) reference electrode described in Section 2-2-1.
- Although the standard electrode potential of Ag/Ag⁺ electrodes varies greatly depending on the solvent, the redox potential of ferrocene is relatively insensitive to solvent effects. Therefore, potential correction using ferrocene is commonly employed.
- Common supporting electrolytes:
As supporting electrolytes that dissolve in organic solvents and have large ions, tetraalkylammonium salts such as tetrabutylammonium perchlorate (TBAP), tetrabutylammonium tetrafluoroborate (TBATFB), and tetrabutylammonium hexafluorophosphate (TBAHFP) are commonly used. - Notes on solvent selection:
When selecting a solvent, it must not only dissolve the analyte but also dissolve its oxidation or reduction products, dissolve the supporting electrolyte to provide sufficient conductivity, and remain stable over a wide potential window. In addition, when the analyte undergoes oxidation or reduction to form ionic species, the interactions between those ions are strongly influenced by the dielectric constant of the solvent. In solvents with a low dielectric constant, ion pairs or aggregates tend to form, which can affect electrochemical behavior; therefore, careful consideration is required. - Recommended organic solvents:
Using solvents with a high dielectric constant is advantageous for sufficiently dissociating the supporting electrolyte and minimizing solution resistance. Commonly used solvents include acetonitrile (AN), propylene carbonate (PC), dimethyl sulfoxide (DMSO), and dimethylformamide (DMF). However, DMF and dichloromethane (DCM) are not recommended for use with Ag/Ag+ electrodes due to issues related to their reactivity with silver ions and solubility.
3-3 Precautions for reference electrodes in epecial environments
Reference electrodes sold by BAS are designed for use at room temperature (25 ℃). Operation outside this temperature range is not guaranteed. When measurements must be performed under non-standard conditions, countermeasures such as isolating the reference electrode body from the measurement solution using a salt bridge or a double-junction chamber are recommended.
Because the Nernst equation, which determines the reference electrode potential, depends on absolute temperature (T), temperature control is extremely important. When measurements are carried out at low temperatures, the solubility of salts in the internal solution of the reference electrode may gradually decrease, potentially leading to salt crystallization.
3-4 Reference electrode selection flowchart: Choose the optimal electrode by simple questions
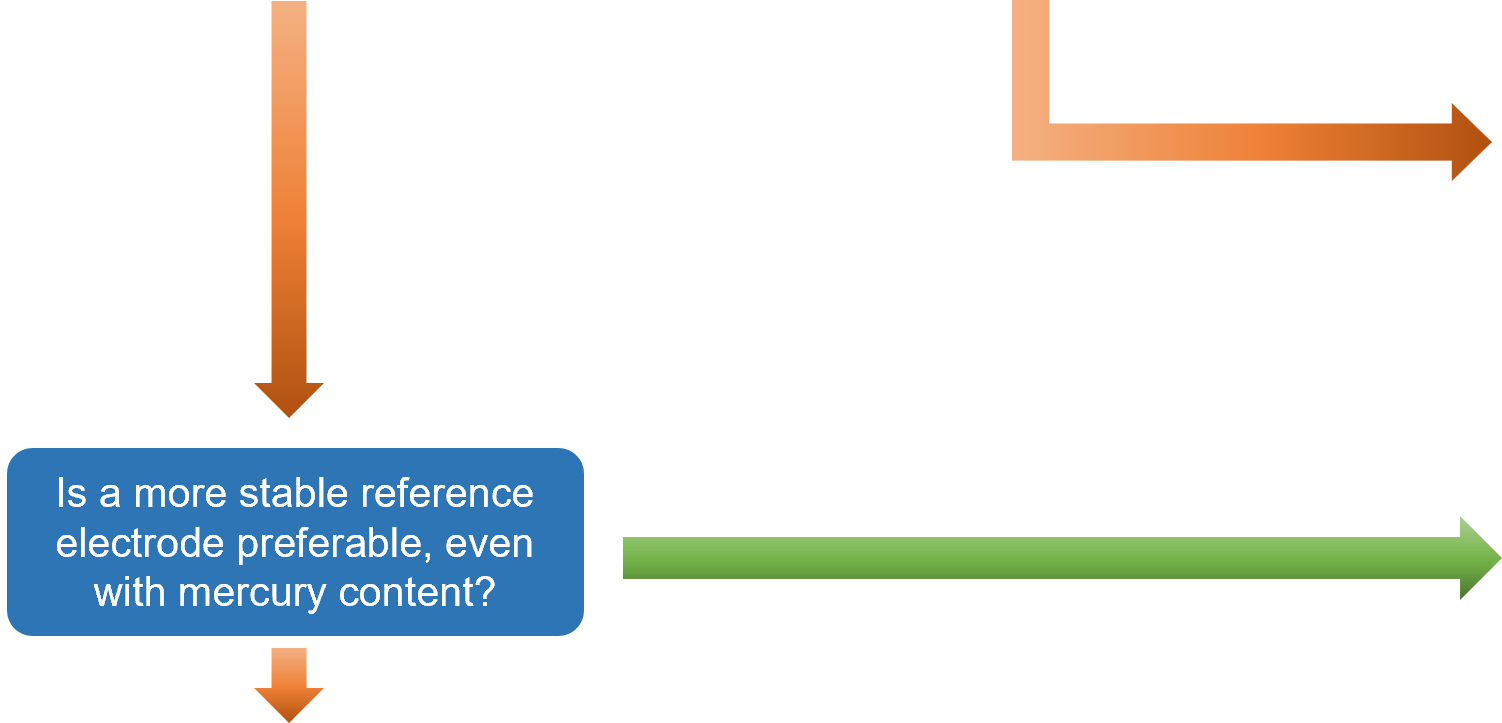
Based on the explanations provided in Sections 3-1 to 3-3, a flowchart has been prepared to guide users to the recommended reference electrode simply by answering YES or NO questions. This flowchart is particularly useful for users selecting a reference electrode for the first time.
After selecting an electrode, clicking on the corresponding "RE-⁎⁎" item will take you to the product page. In some cases, using a double junction chamber, such as with the simplified reversible hydrogen electrode kit (RHEK), can expand the range of applicable measurement conditions.
Chapter 4: Common mistakes and how to prevent them
4-1 Error caused by voltage drop (E = IR): Influence of solution resistance on measurement results~
As explained in Chapters 1–2, in a potentiostat the reference electrode controls the potential of the working electrode. However, if there is a distance between the reference electrode and the working electrode, an uncompensated solution resistance (Ru) exists between them. When current (i) flows through this resistance, a voltage drop (i × Ru) occurs. As a result, the actual potential applied to the working electrode becomes lower than the potential set on the potentiostat.
To avoid this problem, use a potentiostat equipped with an IR compensation function (such as the ALS600 series). Peak broadening caused by IR drop resembles phenomena observed when the electron transfer rate (ks) is slow (irreversible or quasi-reversible systems). If this effect is not properly evaluated and compensated, the experimenter may incorrectly conclude that the electrochemical reaction is slow (low reversibility), even though the system actually has a fast electron transfer rate.
4-2 Electrode deterioration and clogging: Causes and countermeasures for noise and abnormal potential shifts
Before conducting experiments, always confirm that the reference electrode to be used meets its original standard potential. A potentiostat only controls the potential of the working electrode relative to the reference electrode, so it cannot judge the quality of the reference electrode itself.
Please refer to the support page for methods to check reference electrodes. Reference electrodes sold by BAS include recommended storage methods for each electrode type; follow these instructions carefully. If the liquid junction dries out even once, the electrode may become unusable.
If the liquid junction becomes clogged, small noise may appear in the voltammogram. Noise is often assumed to come from the environment (external noise), but the reference electrode itself can also be a source of noise. In addition, if bubbles adhere to the tip of the reference electrode or are present inside the glass tube near the tip, potential control may fail. In such cases, a large potential may be applied to the working electrode, potentially damaging it. Therefore, always check for bubbles before measurement.
4-2 Do not mix aqueous and non aqueous sample: Prevent measurement rrrors due to liquid junction potentials
Do not use aqueous reference electrodes in non aqueous (organic solvent) systems. A large liquid junction potential arises between water and organic solvents, making it difficult to determine accurate electrode potentials.
Chapter 5: Quality and traceability supporting reliability
5**5-1 Product performance and inspection check: Data acces via serial number
At BAS, the potential of each individual reference electrode is measured, and only electrodes that meet our internal q5uality standards are assigned a serial number and shipped.
The greatest benefit to our customers is the assurance of a stable potential reference, which is essential for accurate and reliable electrochemical measurements.
We have established a traceability system that allows customers to access the measurement data obtained during electrode inspection by entering the serial number printed on the electrode.
This system is available no only for reference electrodes but also for nearly all working electrodes sold by our company, and the data can be viewed from this page.
Please select the electrode type from the pull-down menu and enter the manufacturing number, consisting of two alphabetic characters followed by a three-digit number.
Chapter 6: Maintenance and troubleshooting for long-term use
6-1 Reference electrodes used in electrochemical measurements
Reference electrodes are standard electrodes required to exhibit a stable potential. However, with continued use, surface degradation or intrusion of sample solution into the electrode can occur, causing gradual deterioration and loss of the original potential.
6-1-1 Potential verification and criteria for electrode replacement
Method for checking potential
Measure the potential difference between a new reference electrode and the reference electrode in use. Electrochemically, this corresponds to an open-circuit potential measurement. Attach the new reference electrode to the reference electrode clip and the electrode to be tested to the working electrode clip, then measure the potential difference. When comparing new aqueous reference electrodes of the same type, the potential difference typically falls within 5 mV.
Replacement criteria
If the potential deviation of a routinely used reference electrode exceeds ±20 mV from literature values, replacement is recommended. Please refer to the relevant page for details.
6-1-2 Storage and liquid junction precautions (Preventing drying of the liquid junction)
Proper storage is essential to maintain the stability of reference electrodes.
- Cleaning: After use, rinse off any adhered sample solution with distilled water or the organic solvent used.
- Storage: To prevent drying of the ion-permeable glass or ceramic liquid junction at the electrode tip, store the electrode immersed in the recommended storage solution according to the manual, in a cool and dark place. BAS sells the RE-PV reference electrode storage bottle.
- Liquid junction: If the liquid junction dries, salt crystallization may cause damage. Salt precipitation also greatly increases impedance, reducing potentiostat response speed, destabilizing the control system, increasing noise susceptibility, and causing potential shifts. During long-term storage, periodically replace the storage solution and check the electrode potential.
6-2 Precautions for each type of reference electrode
6-2-1 Silver/Silver chloride reference electrodes
(Ag/AgCl types: RE-1CP, RE-1BP, RE-1B)
These electrodes are mainly intended for aqueous samples. As shown in Equation 1 of Chapter 1-3, the chloride ion concentration in the internal solution greatly affects potential shifts, so proper concentration management of the storage solution is necessary. Aqueous reference electrodes cannot be used in non-aqueous solvents. Although not recommended, if unavoidable, a salt bridge must be used.
6-2-2 Mercury-Based Reference Electrodes
(RE-2BP, RE-2CP, RE-61AP)
These electrodes are also primarily intended for aqueous samples. They consist of mercury and mercury chloride (calomel electrode: RE-2BP), mercury sulfate (RE-2CP), or mercury oxide (RE-61AP). Their use has declined in recent years due to environmental concerns. BAS products use polymethylpentene resin containers to improve resistance to external impact.
The RE-61AP is designed specifically for strong alkaline conditions and can use up to 6 M KOH as the internal solution (not included). When filling the solution, wear protective equipment and avoid trapping air bubbles.
6-2-3 Reversible Hydrogen Electrode (RHE)
This electrode uses an internal solution with the same pH as the measurement solution. According to the Nernst equation, its potential decreases by approximately 59 mV relative to the standard hydrogen electrode (SHE) for every increase of one pH unit, an important characteristic unique to this electrode.
BAS RHEs employ a method that temporarily stores hydrogen, requiring preparation according to the manual each time they are used. When combined with a double-junction chamber kit, they can be used not only in acidic solutions but also in neutral solutions.
Chapter 7: Summary on reference electrodes
7-1. The key to accurate experiments: Maintaining a stable "ruler"
A reference electrode functions like a "ruler" that provides a stable reference potential in electrochemical measurements.
If this ruler becomes inaccurate, the reliability of all measurement results is compromised. Therefore, it is essential to regularly check the electrode potential and to store the electrode properly to prevent drying of the liquid junction.
In addition, accurate experiments require a proper understanding of the characteristics specific to each type of electrode, such as the use of mercury or the pH dependence of the Reversible Hydrogen Electrode (RHE).
7-2. Advantages of choosing BAS products
- Provision of high-quality electrodes with excellent potential stability
- Ability to verify inspection data at the time of shipment (customers can personally confirm product quality through our traceability system)
- A wide range of electrodes offering both versatility and specialized performance
- Extensive educational and technical resources on electrochemistry, including "Basics for who are starting electrochemistry", "Electrode Handbook", "BAS Movie Library", and "Electrochemical Glossary".
⁘ Inquiry contacts by purpose
We provide dedicated contact points tailored to different customer needs, including "Product Q & A" and "Manual downloads," to ensure prompt and appropriate support.