Part III: Electrode Potential Equilibrium and Electrode Reaction Kinetics (3)
♦ Standard rate constants
Consider a very special case, such as CR = CO = 1 (mol dm-3) (the standard state for concentration standards), where the reaction is at equilibrium. (Because at equilibrium, the surface concentration equals the bulk concentration.) At this point, the reaction rates from Equations (4) and (5) can be expressed as Equations 15 and 16.
When the reaction reaches equilibrium, the forward and reverse reaction rates are equal (Equation 17). This yields Equation (18). Since it is at equilibrium and the rates are equal, if we express it as Equation (19), This k0 is the standard rate constant. When the reaction reaches equilibrium, the forward and reverse reaction rates are equal (Equation 17), yielding Equation (18). Because it is in equilibrium and the rates are equal, if we express it as Equation (19), this k0 is the standard rate constant.
k0 Standard rate constant
Since the equilibrium potential Eeq equals the molar potential E0, equation (9) can be expressed as equation (20).
Substituting equations (19) and (20) into equation (13) yields a reformulation of the oxidation reaction rate constant kox as equation (21). Similarly, substituting into equation (14) yields the reduction reaction rate constant kred.
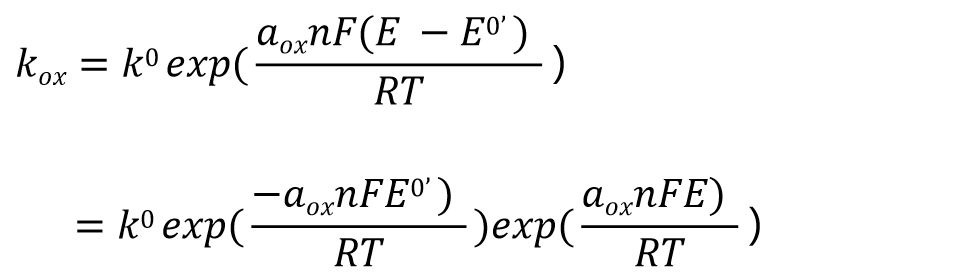
This is the rate constant expression with E0’ as the standard potential.
Comparing equation 21 above with equation (13) for the general equilibrium potential, kox is expressed as equation 22, further transformed into equation 23, and ultimately derived as equation 24.
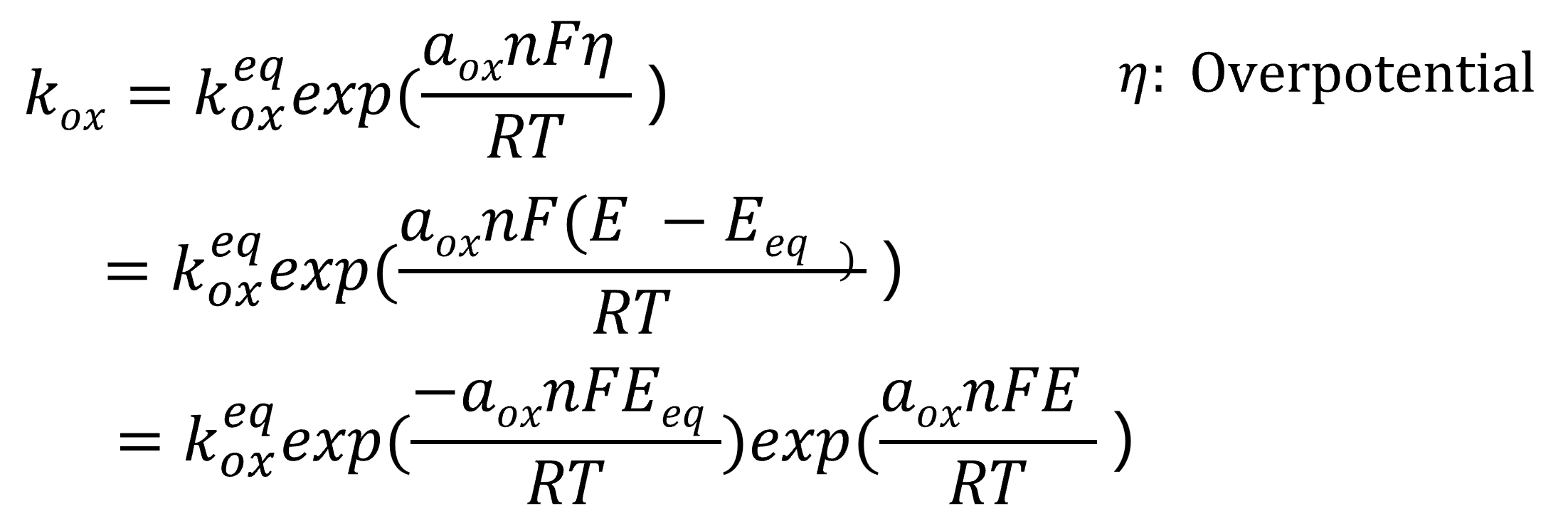
(22)
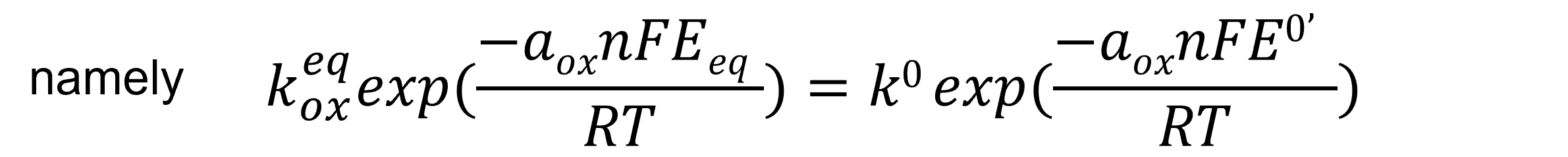

By plotting the equilibrium reaction rate constant keq at the equilibrium potential of oxidation or reduction reactions against applied overpotential, the relationship between the reaction rate constant and potential is obtained (Figure 3).
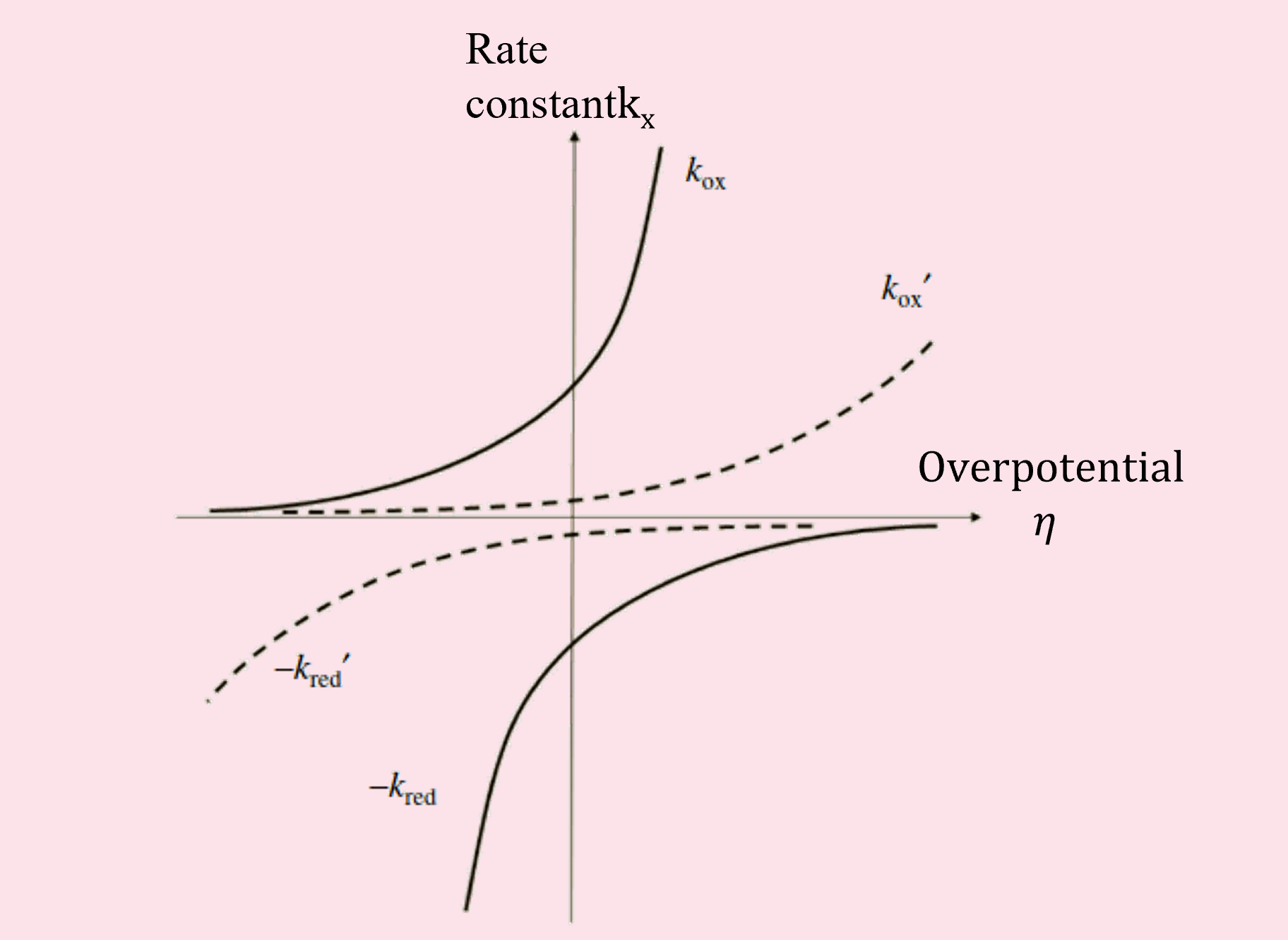
Fig. 3 Dependence of reaction rate constants on potential.
The effects of the overpotential to the large keqox (keqred) reaction (solid line) and the small keqox (keqred) reaction (dashed line) are different. To make it easier to imagine the current, draw kred as a negative number.
The solid line in the figure represents the dependence of the reaction rate constant for oxidation or reduction reactions on potential when the equilibrium reaction rate constant keq is large. The dashed line represents the dependence of the reaction rate constant on potential when keq is small.
The magnitude of the keq coefficient is determined by the nature of the electron transfer reaction rate, which varies with the type and conditions of the reaction. In reactions with a large keq, applying a slight overpotential (low polarity) to the electrode causes a significant change in reaction rate. Conversely, in reactions with a small keq, the reaction cannot proceed unless a large overpotential is applied.

