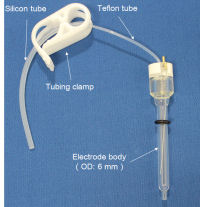
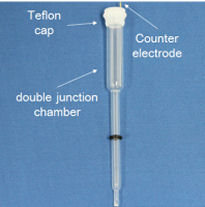
Reversible Hydrogen Electrode kit
It is a reversible hydrogen electrode that achieves good potential stability as a reference electrode for electrochemical measurements by generating and capturing hydrogen gas through electrolysis of an acidic solution.
It makes unnecessary the complicated and dangerous set-up of the hydrogen cylinder and avoids the large volume of hydrogen gas exhaustion, even though it has a good potential stability.
By using a double junction chamber, the electrode can be used with solutions other than acidic solutions.
※ H2G1 Portable Hydrogen Generator, which can be easily filled with hydrogen gas without a potentiostat, is also available.
Troubleshooting
- Specifications
- RHE kit content
- Double jonction chamber kit content
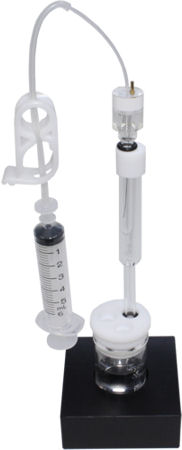
- Setup
- Experimental data
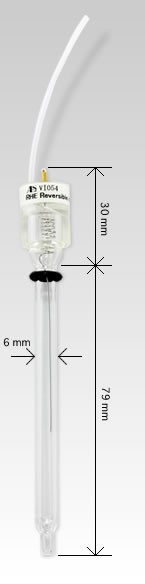
RHE Specifications
| Reversible Hydrogen Electrode (Cat No. 013598) | |
 |
|
| Length | Approximately 109 mm |
| Diameter | Head: 14 mm Neck: 6 mm |
| Temperature range | 5 deg C to 60 deg C |
| pH range (using with Double junction chamber) |
0 to 2 0 to 9 |
Double junction chamber specifications
| Double junction chamber (cat#13376) | |
 |
|
| Length | 140 mm |
| Diameter | Head: 12 mm Neck: 6 mm |
Reversible hydrogen electrode kit
The Reversible hydrogen electrode (RHE) kit can be used without Double junction chamber when strong acid (<pH 2, such as 0.5 mol/L sulfuric acid) is used as an electrolyte solution.
Double junction chamber kit
The Double junction chamber kit is used when the electrolyte solution is a weak acid, neutral solution, solution containing scum etc.
Manuals are available for download here.
Set up of the electrode
RHEK Reversible hydrogen electrode kit only
RHEK Reversible hydrogen electrode kit can be used without "Double junction chamber" when the electrolyte solution is strong acid (<pH2),
such as 0.5 mol/L sulfuric acid.
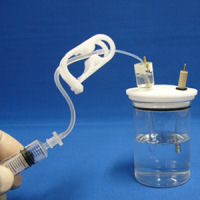 |
Set the RHE Reversible hydrogen electrode and the counter electrode to the electrochemical cell, which contains the strong acid solution. "Disposable syringe" is attached to the open end of "Silicone tube". After confirming whether "Tubing clamp" is released, pull the plunger of the syringe to fill the solution in the electrode. Please check if the liquid level in Silicone tube reaches to the white arrow and there is not any air bubbles in the electrode by looking before closing the tubing clamp. |
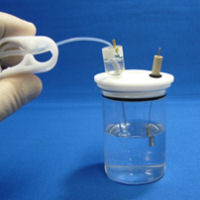 |
Close the tubing clamp, after checking that there is no air bubbles in the electrode. And then, the syringe is detached from the silicone tube. |
 |
Set the potentiostat next to the electrochemical cell. Connect the electrode lead for working electrode (green covering) to the RHE. Please make sure that the leads for reference (white covering) and counter electrodes (red covering) are connected together. |
 |
Electrolysis is done by amperometric i-t technique or chronoamperometric technique during the charge value gets to about two coulombs. In this case, please make sure setting the minus (-) applied potential based on counter electrode. When you use 1.2 mol/L hydrochloric acid as electrolyte solution, the applied potential is set as -3 V and the period of electrolysis would be about 5 minutes. When you use 0.5 mol/L of sulfuric acid, -3 V of applied voltage, the electrolysis time would be about 9 minutes. Caution:The current during the electrolysis is possibly above the current limit of your potentiostat. Please check the specifications of your instrument first before the electrolysis. If pH of the solution is over 2. It would take time for electrolysis. In this case, the applied potential should be changed to the negative direction. Do not touch the tubing clamp or the electrode before ending the experiment. Otherwise, you may fail your experiment. |
 |
The electrochemical test is done by connecting the reference electrode lead to the contact pin of RHE. |
RHEK with Double junction chamber kit
Use this chamber when weak acid, neutral solution, solution containing scum, etc., is used.
 |
Fill the chamber with a strong acid, such as HCl, HClO4, H2SO4, etc.. The RHE would be similar to Standard Hydrogen Electrode (SHE) by using 1.2 mol/L Hydrochloric acid as electrolyte solution. Make sure that there is not air bubbles in the solution, to eliminate the bubbles tap gently the chamber with the finger. |
 |
Fit the "Double junction chamber cap" on the chamber with a counter electrode and hold it by something like test tube stand or voltammetry cell. Do not touch the bottom of the tip of the chamber during the preparation. |
 |
Set the RHE, which was previous preparing with a syringe and tubing clamp setting (see the picture at the left), into the PTFE cap hole. Check if the electrode liquid junction is immersed to the solution. |
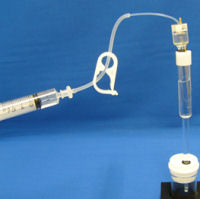 |
Loose the clamp and then pull the plunger of the syringe to take acid solution in the electrode while pushing the electrode down in the chamber securely. Please confirm the condition whether the solution reaches the position marked by white arrow and there is no air bubbles in the solution. |
 |
Make sure closing tubing clamp. Then, set your potentiostat next to the electrochemical cell. Connect the electrode lead for working electrode (green covering clip) with the contact pin of your RHE. Please make sure connecting your electrode leads for reference (white covering clip) and counter electrodes (red covering clip) with the contact pin of a counter electrode together. |
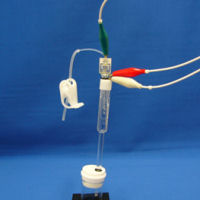 |
Electrolysis is done by amperometric i-t technique or chronoamperometric technique during the charge value gets to about two coulombs. In this case, please make sure setting the minus applied potential based on counter electrode. When you use 1.2 mol/L hydrochloric acid (HCl) as electrolyte solution, The applied potential is set as -3 V and the period of electrolysis would be about five minutes. Caution:The current during the electrolysis is possibly above the current limit of your potentiostat. Please check the specifications of your instrument first before the electrolysis. If pH of the solution is over 2. It would take time for electrolysis. In this case, you should change the applied potential to the negative direction. Never touch the tubing clamp or the electrode before ending the experiment. Otherwise, you may fail your experiment. |
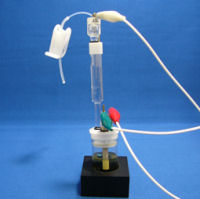 |
Replace the test tube stand with your electrochemical cell containing test sample. Place the working electrode and counter electrode. And then, connect among your potentiostat and the electrodes to measure your sample. |
RHEK (Reversible hydrogen electrode kit) was tested for the evaluation, where the stability of the potential in function of the time and temperature was tested and compared with the reference against SHE. Also, the potential difference between RHEK and line up reference electrode was measured.
Related product
H2G1 Portable Hydrogen Generator, even without a potentiostat, makes easily to charge the hydrogen gas into the RHEK Reversible hydrogen electrode kit.